Treatment Resistant Depression
Treatment-Resistant Depression/TRD and TMS
Why TMS? Transcranial Magnetic Stimulation is one of the most effective treatments for difficult depression available in the U.S. The typical TMS patient has Treatment Resistant Depression or TRD, which has recently been defined as people who have failed to respond to 2 or more antidepressants in the same depressive episode. There has been several other definitions and most insurance company require a failure of 4 antidepressants to achieve a response but experts in the field are now reaching consensus that TRD is failing 2. That said, our typical TMS patient has failed 6-10 antidepressants and several have failed to respond to ECT or shock treatment. In 2010 the American Psychiatric Association published a consensus guideline for treating major depression and in essence said that after 1 antidepressant failure, all options should be on the table including TMS.The data on achieving full remission of chronic depression is not promising with medications. In 2006 the first large, multi-site, several year study of typical outpatients with depression was completed, with results published in 2008. The STAR*D trial was the Sequenced Treatment Alternatives to Relieve Depression. It was funded by the National Institute of Mental Health, not sponsored by pharmaceutical companies. Overall, 4,041 participants from 41 psychiatric and primary care centers were enrolled into Level 1 of the STAR*D study. The average age was 41 years, with about 13 years of education; 64 percent were female, 36 percent were male. Practitioners in the clinical sites identified people with depression who were either already in treatment or were about to be treated at the clinics and offered them the opportunity to participate in the study. Media advertising was not used to attract participants to the STAR*D study. The participants in STAR*D had moderate to severe depressive symptoms, with 75 percent having two or more episodes of depression in their lifetime and 25 percent reporting that their current episode of depression had continued for at least two years. More than one third of the participants were under age 18 when they first experienced depression and participants had an average of three or more general medical conditions.
The results showed that the average remission rate decreased with each level in the study. These were open-label (both the subject and the doctor knew what medications were being administered) and the doctors could select based upon clinical judgement but had to adhere to these levels.
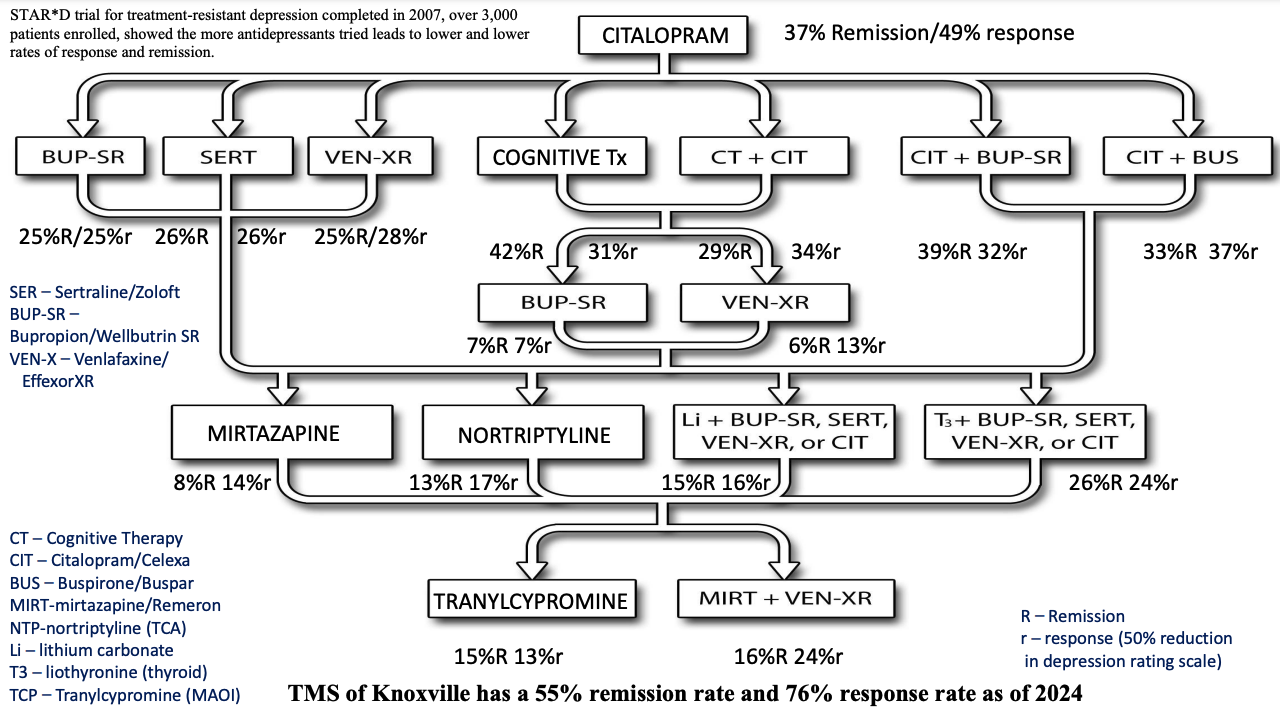
With the first antidepressant citalopram or Celexa, people were allowed to be on it up to 14 weeks or unless side effects forced a discontinuation. Remission rates averaged 37% in this first level. If remission was not achieved, the patients went to level 2 which included switching to other antidepressants, cognitive therapy alone, cognitive therapy plus citalopram, or augmenting/boosting the antidepressant effect by adding bupropion/Wellbutrin or buspirone/Buspar. The remission rate dropped to 31% in this level. By level 3, there was a precipitous drop off by switching to either mirtazapine/Remeron or nortriptyline/Pamelor or augmenting venlafaxine/EffexorXR or citalopram/Celexa with either lithium or T3 thyroid medication/Cytomel. The average remission rate in this level dropped to 14% and by level 4, even using an MAO inhibitor tranylcypromine/Parnate or combining mirtazapine with venlafaxine dropped to 13-15%. Also dropouts increased to 90% by level 4 and side effects increased.
Relapse rates after 1 year strongly correlated with number of failed trials. The likelihood of both remitting and maintaining remission for 1 year at each level (probability of remitting x probability of staying well for following year) was 26% for level 1, 14% for level 2, 5% for level 3, and 3% for level 4.
At the time of this study, augmentation with aripiprazole, quetiapine, or brexpiprazole were not yet available or FDA approved as augmentation agents for major depressive disorder so they were not included.
 As you can clearly see, TMS in our clinic clearly beats all 4 levels of medications
As you can clearly see, TMS in our clinic clearly beats all 4 levels of medications
Now does this mean anyone with major depression who’s tried 2 antidepressants has a very low chance of remission? Not necessarily. Using the proper dose for the proper duration is critical. For example, sertraline/Zoloft dosing range is 50 to 200mg a day. If someone was on 50mg for 3 weeks and felt no better and stopped it, that is not a failed try. One would need to be on doses up to 200mg a day for at least 8 weeks to deem it a failure. Nor would someone who skipped doses qualify. Nor someone actively abusing alcohol or drugs while on it. For a list of current antidepressants and appropriate dosing range, see pages 12-13 of the Treating Major Depressive Disorder – A Quick Reference Guide
References
1. Conway CR, George MS, Sackheim HA: Toward an Evidence-Based, Operational Definition of Treatment-Resistant Depression, When Enough is Enough. JAMA Psychiatry, Jan 2017, vol 74, #1, pp. 9-10
2. Questions and Answers about the NIMH Sequenced Treatment Alternatives to Relieve Depression (STAR*D) Study — Background https://www.nimh.nih.gov/funding/clinical-research/practical/stard/backgroundstudy.shtml
3. Treating Major Depressive Disorder – A Quick Reference Guide found here: http://psychiatryonline.org/pb/assets/raw/sitewide/practice_guidelines/guidelines/mdd-guide.pdf
